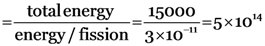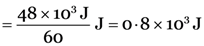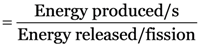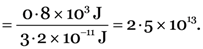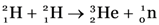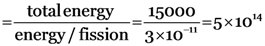Sources of energy Worksheet-11
-
How were fossil fuels formed? How these fossil fuels were made?
-
Name the places of our Country where fields of natural gas are found. Why is it called a clean fuel? Give two reasons.
-
Define the term 'fuel'. What are primary and secondary fuels? To which class of fuel do the following belong :
Coke, Wood, Petroleum, LPG.
-
48 kJ of energy is produced per minute in a nuclear reactor. Calculate the number of fissions which would be taking place in the reactor per second, if the energy released per fission is 3.2 × 10–11 J.
-
What are the disadvantages of using dry wood as domestic fuel?
-
Give one example of a nuclear fusion reaction. Describe one method for making such reactions possible.
-
Which type of nuclear process is currently used in nuclear electricity generators? Give one example each for the substances used in this context as (i) coolants moderators and (iii) nuclear fuel?
-
Define a nuclear fusion reaction. Describe the conditions for the occurrence of a nuclear fusion reaction.
-
What is the cause of release of unusually large energies in nuclear fission reactions? How is the energy per fission calculated?
-
In one fission of uranium, 3.2 × 10–11 J of energy is made available. Calculate the total number of fissions necessary per second to generate power of 15 kW.
Answer:
-
Fossil fuels were formed due to high pressure, heat and the action of anaerobic bacteria on fossils for million of years. These fuels were made form the fossils of plants and animals buried deep in the Earth.
-
Tripura, Jaisalmer, off-shore area of Bombay, Krishna-Godavari Delta.
Natural gas is a clean fuel as :
(i) it does not produce harmful gases on burning,
(ii) It burns without smoke and does not leave any residue.
-
A material which produces heat on combustion is called a fuel.
Primary Fuels. Fuels which are used directly to produce heat are called primary fuels.
Secondary Fuels. Fuels which are manufactured by chemical process using primary fuels are called secondary fuels.
(i) Wood and Petroleum are primary fuels and (ii) Coke and LPG are secondary fuels.
-
Total energy produced/minute = 48 kJ = 48 × 103 J
Energy produced/second 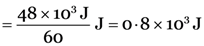
Energy released/fission = 3.2 × 10–11 J.
Number of fissions/second
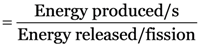
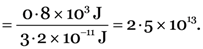
-
(i) Wood produces lot of smoke on burning.
(ii) It leaves lot of residue after burning.
-
One example of a nuclear fusion reaction is when two deuterons fuse together to form He-3 nucleus and a neutron with the emission of a large amount of energy
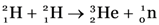
This reaction is possible at extremely high temperatures (≈ 107 K) and pressures (≈ 107 Pa).
-
In nuclear electricity generators, the process used currently involves the fission of U-235 nuclei by bombarding them with thermal neutrons.
Water is one of the coolants ; graphite is one of the moderators and enriched uranium is one of the nuclear fuels.
-
A nuclear fusion reaction involves combining of two or more lighter nuclear to form a heavier nucleus with the liberation of a large amount of energy.
Conditions :
(i) Temperature of fusing nuclei should be raised to 107 K at pressure of millions of Pascal.
(ii)The fusing nuclei be accelerated to high speeds using particle accelerators.
-
In nuclear fission reactions, certain mass disappears. This is called mass defect (Δm). This mass appears in the form of energy according to Einstein equation, E = (Δm)c2, where c is velocity of light in vacuum. To calculate energy per fission, we sum up the masses on left hand side of the fission reaction; and also on the right hand side of the fission reaction .The difference in masses on the two sides of the reaction gives us mass defect (Δm). Usually, this mass defect is in atomic mass unit (u). As 1 u = 931 MeV, we can calculate energy released/fission (in MeV) by multiplying (Δm) by 931.
-
Energy released/fission = 3 × 10–11 J
Total energy required = 15 kW = 15000 w = 15000 J/s.
Number of fissions