Atoms and Molecules Worksheet-3
A. NaO and Na2S B. Na2O and NaS2
C. NaO2 and Na2S D. Na2O and Na2S
A. Aluminium sulphate B. Aluminium sulphite
C. Aluminium sulphide D. Aluminium thiosulphate
A. Calcium bicarbonate
B. Calcium carbonate
C. Calcium hydrogen carbonate
A. Potassium sulphite
B. Potassium sulphide
C. Potassium hydrogen sulphate
D. Potassium sulphate
A. 33 u B. 24 u C. 32 u D. 31 u
K = 39 u, C = 12 u and O = 16 u
A. 138 u B. 132 u C. 122 u
(a) 100 grams of sodium (b) 100 grams of iron
(Given : atomic mass of Na = 23u ; Fe = 56u)
A. 100 grams of iron contains more number of atoms
B. 100 grams of sodium contains more number of atoms
C. 100 grams of sodium and 100 grams of iron contain equal number of atoms
A. 63 u B. 36 u C. 60 u D. 66 u
A. 1 mole B. 0.1 mole C. 0.5 mole D. 0.25 mole
A. 0.240 × 1023 particles B. 0.240 × 1023 particles
C. 2.05 × 1023 particles D. 6.022 × 1023 particles
A. 4g of methane (CH4) has more number of molecules
B. 11g of CO2 has more number of molecules
C. Both have same number of molecules
A. Law of conservation of mass
B. Law of constant proportions
C. Law of reciprocal proportions
D. Law of multiple proportions
A. 6.022 × 1023 molecules are present in 1 mole of hydrogen molecule
B. Gram molecular mass of 1 mole of Hydrogen molecule is 2g
C. 2 × 6.022 × 1023 atoms are present in 1 mole of Hydrogen molecules
D. None of these
A. 10 g of sulphur dioxide (SO2)
B. 10g of oxygen (O2)
C. Both have same number of molecules
A. XO2 and XO3 B. X4O2 and X6O2
C. XO2 and X3O6 D. XO and XO3
A. Flask P
B. Both contain same number of oxygen atoms
C. Flask Q
Answer:
Explanation:

Explanation: Aluminium sulphate
Explanation: Calcium carbonate.
Explanation: Potassium sulphate
Explanation: Methyl alcohol (CH3OH)
Molar mass of CH3OH = (1 × atomic mass of C) + (4 × atomic mass of H) + (1 × atomic mass of O)
= (1 × 12u) + (4 × 1u) + (1 × 16u) = 32u
Explanation: Formula unit mass of K2CO3 (Potassium carbonate).
= (2 × atomic mass of K) + (1 × atomic mass of C) + (3 × atomic mass of O)
= (2 × 39u) + (1 × 12u) + (3 × 16u) = 138u
Explanation: (a) 100 grams of sodium
No. of atoms of sodium = Number of moles × (avogadro's number)
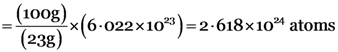
(b) 100 grams of iron
No. of atoms of iron = Number of moles × (Avogadro's number)
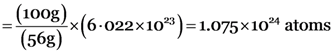
100 g of sodium has more number of atoms
Explanation: Nitric acid, HNO3
Molar mass of HNO3 = (1 × atomic mass of H) + (1 × atomic mass of N) + (3 × atomic mass of O)
= (1 × 1u) + (1 × 14u) + (3 × 16u) = 36u.
Explanation: 22 g of carbon dioxide (CO2)
Molar mass of carbon dioxide (CO2) = (1 × 12u) + (2 × 16u) = 44u
Gram molecular mass = 44g
44 g of carbon dioxide (CO2)= 1 mol
22g of carbon dioxide (CO2) 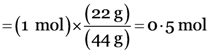
Explanation: 1.0 mole of the element represents particles = 6.022 × 1023
0.25 mole of the element represents particles 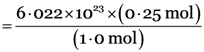
= 1.505 × 1023 particles
Explanation: Number of moles in 4 g of methane (CH4) 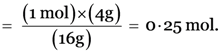
Number of moles in 11 g of CO2 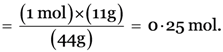
Since both CH4 and CO2 have same number of moles (0.25 mol), they have same number of molecules also.
Explanation: Carbon dioxide consists of carbon and oxygen. Elements combine in a fixed ratio to form a compound. According to the Law of constant proportions the composition of a compound remains the same irrespective of the source. Since the percentage of carbon in each sample is fixed, that of oxygen must be also fixed. This is according to law of constant proportions.
Explanation: All the statements are correct.
Explanation: Number of molecules in 10 g of sulphur dioxide (SO2)
Molar mass of SO2 = 32g + 2 × 16g = 64g
Number of molecules of sulphur dioxide in 64 g of sulphur dioxide = 6.022 × 1023
Number of molecules of sulphur dioxide in 10 g of sulphur dioxide
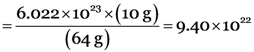
No. of molecules in 10 g oxygen
Molar mass of oxygen (O2) = 32g
32 g of oxygen represent molecules = 6.022 × 1023
10 g of oxygen represent molecules
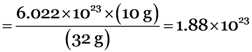
Thus, 10 g of oxygen (O2) has more molecules than 10g of (SO2)
Explanation: Let the element be X
Formula of oxide in which valency of X is 4 = X2O4 or XO2
Formula of oxide in which valency of X is 6 = X2O6 or XO3
Explanation:
1 molecule of oxygen (O2) = 2 atoms of oxygen
1 molecule of ozone (O3) = 3 atoms of oxygen
In flask P : 1 mole of oxygen gas = 6.022 × 1023 molecules
0.5 mole of oxygen gas = 6.022 × 1023 × 0.5 molecules
= 6.022 × 1023 × 0.5 × 2 atoms
= 6.022 × 1023 atoms
In flask Q : 1 mole of ozone gas = 6.022 × 1023 molecules
0.4 mole of ozone gas = 6.022 × 1023 × 0.4 molecules
= 6.022 × 1023 × 0.4 × 3 atoms
= 7.23 × 1023 atoms
∴ Flask Q has a greater number of oxygen atoms as compared to flask P.