Atoms and Molecules Worksheet-2
A. 33 g B. 30 g C. 31.68 g D. 29 g
A. 0.0199 mol B. 0.01 mol
C. 1.015 mol D. 0.009 mol
(i) 46 g of Na (ii) 60 g of Ca
Given gram atomic mass of (i) Na = 23 g (ii) Ca = 40 g.
A. (i)2 mol (ii) 1.5 mol
B. (i)1.5 mol (ii) 1.5 mol
C. (i)2 mol (ii) 2 mol
D. (i)2 mol (ii) 1 mol
(Given : Gram atomic mass of oxygen = 16 g, Gram molecular mass of oxygen (O2) = 32 g)
A. 16g B. 32g C. 8g
A. 27 u B. 108 u C. 81 u
A. 142 u B. 140 u C. 1420 u D. 1400
A. 28 g B. 44g C. 22 g D. 11g
A. 6.022 × 1023 atoms B. 3.022 × 1023 atoms
C. 6.022 × 1030 atoms D. 6.022 × 1028 atoms
A. 16 B. 32 C. 48 D. 64
(i) 10 g of Ca. (ii) 2.3 g of Na
A. (i) 0.5 mole (ii) 0.1 mole
B. (i) 0.25 mole (ii) 0.5 mole
C. (i) 0.75 mole (ii) 0.1 mole
D. (i) 0.25 mole (ii) 0.01 mole
Answer:
Explanation:
Molecular mass of CO2 = atomic mass of C + 2 × atomic mass of O
= (12u + 2 ×) 16u = 44u
gram molecular mass of CO2 = 44g
1 gram molecular of CO2 = 44g
0.72 gram molecular of CO2 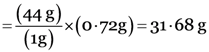
Explanation: 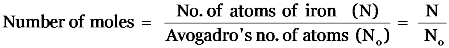
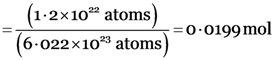
Explanation: (ii) 46 g of Na
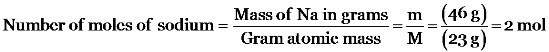
(iii) 60 g of Ca
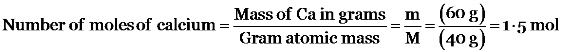
Explanation: 6.022 × 1023 molecules of oxygen (O2)
I mole of a substance contains Avogadro number of particles.
Step I: Calculation of number of gram moles of oxygen.
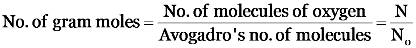
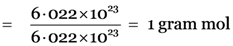
Step II: Calculation of mass of oxygen (O2) molecules.
Mass of oxygen (O2) molecules = Gram molecular mass of oxygen × No. of gram moles of oxygen.
= (32 g) × 1 = 32g.
Explanation: 4 moles of aluminium atoms
Mass of 1 mole of aluminium (Al) atoms = 27 u
∴ Mass of 4 moles of aluminium (Al) atoms 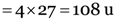
Explanation: Mass of 1 moles of sodium sulphate (Na2SO4) = 142 u
Mass of 10 moles of sodium sulphate (Na2SO4) = 1420 u
Explanation: (i). Calculation of the number of oxygen atoms in 22 g of carbon dioxide (CO2)
Molar mass of CO2 = 12 + 2 × 16 = 44u
1mole of a substance = 1 Gram molecular mass of the substance.
44 g of CO2 = 1 mol
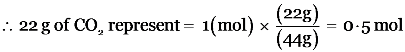
The number of oxygen atoms in 1 mole of CO2 = 2 × N0
= 2 × 6.022 × 1023
∴ the number of oxygen atoms 0.5 mole of CO2 = 2 × 6.022 × 1023 × 0.5
= 6.022 × 1023 atoms.
(ii). Calculation of weight of carbon monoxide (CO)
6.022 × 1023 atoms of oxygen are present in 6.022 × 1023 molecules of carbon monoxide (CO).
According to mole concept 1 mole of a substance contains Avogadro number of particles.
6.022 × 1023 molecules of CO have mass = Molar mass of CO = 12 + 16 = 28 g.
Explanation: One gram atomic mass of a substance contains Avogadro number of atoms i.e. 6.022 × 1023
Explanation: Gram atomic mass of one mole of oxygen atoms = 16 g
∴ Mass of 2 moles of oxygen atoms = 32 g
Explanation: 1 gram atomic mass of calcium = 40 g = 1 mole
10 g of calcium = 0.25 mole
1 gram atomic mass of sodium = 23 g = 1 mole
2.3 g = 0.1 mole