Combustion and flame Worksheet-6
-
An important liquid fuel, used in home is _____.
-
What are the main constituents of biogas and kitchen gas (L.P.G.)?
-
Fuel must be heated to its ____ temperature before it starts burning.
-
Name the gaseous fuels.
-
Make a labelled diagram of a candle flame
-
What name is given to the substances 'which can burn easily?
-
What name is given to the substances which do not burn?
-
When does a substance start burning?
-
Give two examples of inflammable substances.
-
What is a flame?
-
What is meant by rapid combustion?
-
What is meant by calorific value?
-
How is acid rain harmful?
-
Why is respiration referred to as slow combustion?
-
Why does yellow phosphorus catch fire of its own?
Answer:
-
LPG (liquid petroleum gas)
-
Hydrocarbons are the main constituents of biogas and kitchen gas (L.P.G.).
-
Ignition
-
Natural gas, Biogas, CNG
-
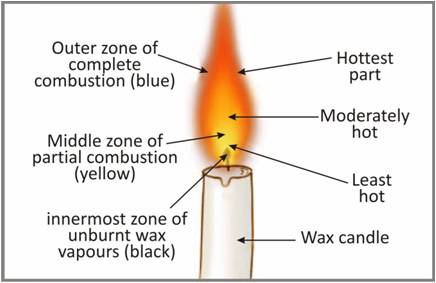
-
Substances which burn in air to produce heat and light are called combustible substances.
E.g.:- wood, coal, charcoal, kerosene, petrol, diesel, liquefied petroleum gas (LPG), compressed natural gas (CNG) etc.
-
Substances that do not burn in air or oxygen to produce heat and light are called non-combustible substance.
Water, sand, glass, and cement are examples of non-combustible substances.
-
A substance starts burning when its ignition temperature is reached.
-
Petrol, alcohol, diesel, Kerosene
-
Flame is the zone of combustion of a combustible substance. Substances which vapourise during burning produce flames.
-
Rapid combustion: In this type of combustion, large amount of heat and light is released in a very short span of time.
Example: Combustion of LPG, which produces heat and light instantly
-
Calorific value of a fuel is defined as the amount of heat produced in kilojoules when one gram of a fuel is completely burnt.
-
Acid rain makes the soil acidic which harms the plants. Acid in the rain also decolourises the historical monuments.
-
Combustion is the process in which a substance reacts with oxygen producing energy in the form of heat and light.
In the process of respiration carbohydrates react with oxygen producing carbon dioxide, water and energy. The reaction in respiration is slow hence it is referred as slow combustion.
-
Yellow phosphorous reacts with oxygen as soon as it is exposed to air producing energy hence it catches fire of its own.
