Acids, bases and salt Worksheet-8
One Word Questions:
What will be the colour of litmus solution when it is added to soda water?
Name the acid present in curd.
What is the colour of phenolphthalein in dil. HCl ?
What will be the colour of litmus paper in solution of bleaching powder in water?
Which gas is evolved when zinc granules react with dil. H2SO4 ?
What is the name of the compound formed when CO2 gas is passed through limewater?
What will be the concentration of H+(aq) ions in the solution when pH = 0?
What is pH of acid rain?
What is the colour shown by universal indicator in neutral medium?
A yellow coloured solution turns reddish pink in acidic solution. What is the substance called?
Name an indicator extracted from Lichen?
Egg shells are made of a compound (A) which when treated with dil. Hydrochloric acid evolves a gas (B). (B) turns lime water milky.
What is the colour of phenolphthalein solution in basic medium?
Acids are stored in _______ containers.
Which gas is evolved when acids react with metal carbonates?
Which non-metal is common to all acids?
Which ion is responsible for basic nature of base?
Bases which are soluble in water are called.
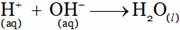 is a
is a
Name a sodium compound which is used for making borax and glass.
Name the constituents of soda-acid type of fire extinguishers.
A substance which removes colour from coloured substances and make them colour less is called.
Answer keys:
Red
Lactic acid
Colourless
Blue
Hydrogen
Calcium carbonate
1
5/6
Green
Indicator
Litmus
A-Calcium carbonate B-Carbon dioxide.
Pink
Glass/Ceramic
Carbon dioxide/CO2
Hydrogen.
Hydroxide ion /(OH–)
Alkali.
Neutralization Reaction.
Sodium carbonate / washing soda
Sodium hydrogen carbonate and sulphuric acid.
Bleaching agent.