Combustion and flame Worksheet-4
Multiple Choice Questions:

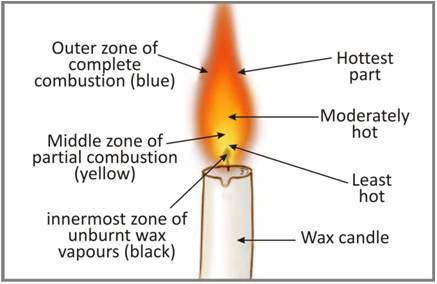
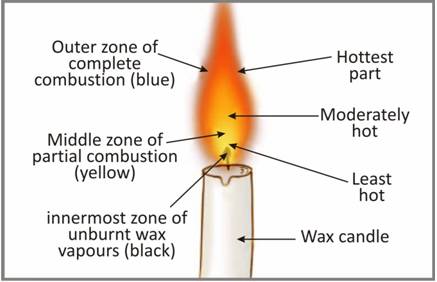
Yellow colour in the flame denotes ______.
A. Zone of complete combustion, luminous
B. Zone of incomplete combustion, luminous
C. Unburnt wax vapours
D. None of these

The inner zone (black) denotes the______.
A. Zone of complete combustion, luminous
B. Zone of incomplete combustion, luminous
C. coolest part of the flame , Unburnt wax vapours
D. None of these
A. The amount of heat produced in kilocalories when one gram of a fuel is completely burnt.
B. The amount of heat produced in kilojoules when one gram of a fuel is completely burnt.
C. The amount of heat produced in joules when one gram of a fuel is completely burnt.
D. The amount of heat produced in calories when one gram of a fuel is completely burnt.
A. more
B. Less
C. Information insufficient to answer
D. None of these
A. CNG B. LPG C. Wood D. Coal
A. Petrol B. Diesel
C. CNG D. None of these

Change in the colour of bunsen flame is due to:
A. Fuel is the same, Oxygen supply is increased
B. Fuel is the same, Oxygen supply is decreased
C. Fuel is changed and Oxygen supply is increased
D. Fuel is changed and Oxygen supply is decreased
increase in oxygen supply the colour of fuel becomes blue.
A. Water B. CO2 C. Oxygen D. Hydrogen
A. Low B. High C. Moderate D. Zero
A. Incomplete combustion of wood
B. Complete combustion of wood
C. Complete combustion of charcoal
D. Combustion of CNG
Answer:
Explanation: Calorific value of methane is 55 KJ/g and of LPG is 50 KJ/g
Explanation: Among the given fuels LPG has highest calorific value and it does not produce carbon monoxide and carbon particles also. Hence it is the best domestic fuel.
CNG is used as fuel in automobiles.
Explanation: The flame produced by complete combustion of fuel is blue in colour. For complete combustion excess of oxygen is required. So with the
Explanation: Water cannot be used as fire extinguisher in electric fires and also fire caused by oil. In other type of fires water is used as fire extinguisher.
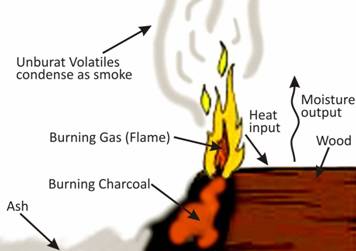
Due to incomplete combustion of wood/charcoal carbon monoxide and carbon particles are produced.