Carbon and its compounds Worksheet-3
Fill in the blanks:
Valency of carbon is _____.
__________ bonds are present between carbon atom in unsaturated compounds.
There are _____ hexagonal rings in Buckminster fullerene.
______ is the general name of the class of compounds having general formula CnH2n+2.
_______ is the homologous series of the organic compounds of formula C18H36.
The name of the first member of the compound of the homologous series of general formula CnH2n+1Cl is ______.
The saturated hydrocarbons generally burn in air with blue and non-sooty flame because the percentage of carbon is _______ in them.
Multiple-Choice Question:
(A) CO2 (B) CO,CO2 (C) H2,CO2 (D) N2,H2,CO
(A) Hexene (B) Hexane (C) Hexyne (D) None of these
(A) C8H17 (B) C8H19 (C) C8H16 (D) C8H14
(A) CH3 (B) C2H2 (C) CH2 (D) CnH2n
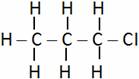
(A) Ethyl chloride (B) Chloropropane
(C) Chloroethane (D) Chlorobutane
Answer key
4/ four
Double/triple
20/ twenty
Alkanes/Alkane
Alkene
Methyl chloride/Chloro methane
Comparatively less/ less
(A)
(C)
(C)
(C)
(B)