CHEMICAL FORMULAE
-
A chemical formula represents the composition of a molecule of the substance in terms of the symbols of the elements present in the molecule.
-
While writing the formula of a molecular compound the less electronegative non-metal element is written on the left hand side whereas the more electronegative non-metal element is written on the right hand side.
-
In naming the molecular compounds, the name of less electronegative non-metal is written as such but the name of more electronegative non-metal is changed to have the ending ‘ide’.
-
When there are more than one atoms of an element in a molecular compound, then ‘the number of atom’ is indicated by using appropriate ‘prefixes’ in the formula.
-
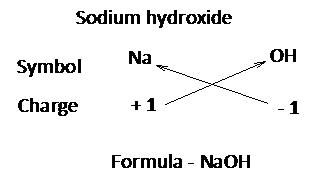
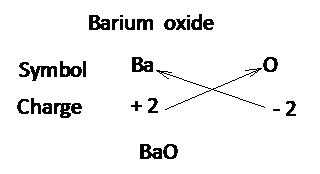
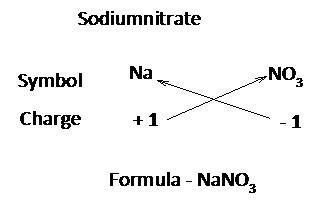
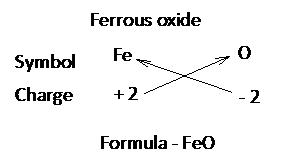
While writing the formula of ionic compounds as a convention, the cation (positive ion) is written on the left hand side and the anion (negative ion) is written on the right had side.
-
The number of cations and anions is adjusted in such a way that the total number of positive valencies of cations becomes equal to the total number of negative valencies of anions
Formulae of compounds:
-
The chemical formula of a compound is a symbolic representation of compound which tells about the number of atoms of each element present in the compound.
For example:
H2O 2 atoms of Hydrogen and 1 atom of Oxygen
SO2 2 atoms of Sulphur and 2 atoms of Oxygen
NH3 1 atom of Nitrogen and 3 atoms of Hydrogen
HCl 1 atom of Hydrogen and 1 atom of chlorine
C2H5OH 2 atoms of carbon, 6 atoms of Hydrogen and 1 atom of oxygen
CCl4 4 atoms of chlorine and 1 atom of carbon
Writing chemical formula of a molecular compound:
Step 1:
Write the symbols of the elements present in the compound.
Example:
Aluminium chloride
Aluminium [Al] Chlorine [Cl]
Step 2:
Write the valency of each element.
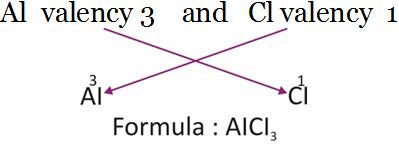 (Cross over of valency of combining atoms)
(Cross over of valency of combining atoms)
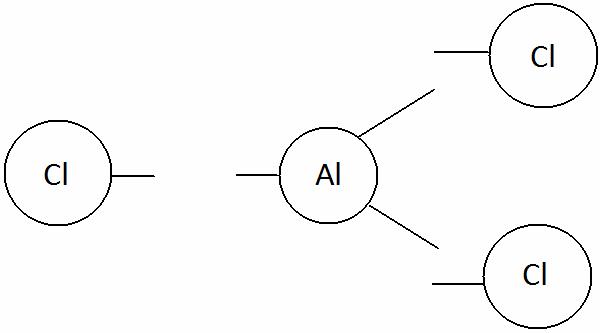
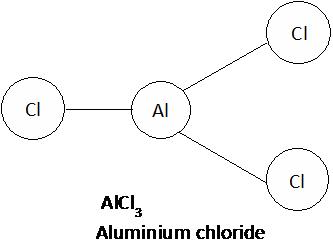
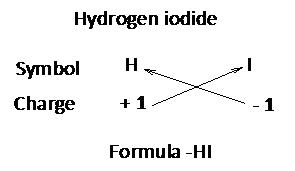
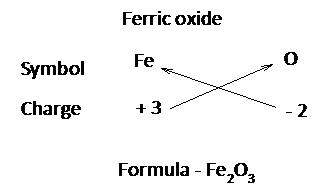
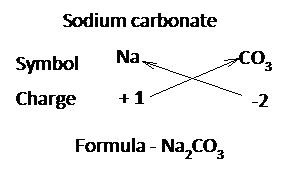
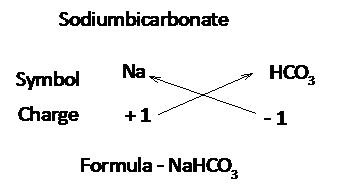
 (Cross over of valency of combining atoms)
(Cross over of valency of combining atoms)








