Carbon Compound and Petroleum Worksheet-2
2CaC2 + 2H2O → (A) + (B)
If (A) is involved in the neutralization reaction with acid, then (B) may be
(a) C2H2 (b) CH4 (c) Ca(OH)2 (d) CaH2
(a) CaCO3 which is soluble in H2O
(b) CaCO3 which is insoluble in H2O
(c) CaHCO3 which is soluble in H2O
(d) CaHCO3 which is insoluble in H2O
(a) Non-renewable, renewable
(b) Renewable, non-renewable
(c) Renewable, renewable
(d) Non-renewable, exhaustible
(a) above the petroleum oil
(b) below the petroleum oil
(c) along with the petroleum oil
(d) In the environment
(a) Black gold (b) Yellow gold
(c) Green gold (d) Blue gold
(a) wood and coal (b) LPG and cow dung
(c) petroleum and coal (d) natural gas and coal
(a) Coal gas (b) Marsh gas
(c) Natural gas (d) Gas carbon
(a) Anthracite (b) Bituminous
(c) Peat (d) Lignite
(a) C4H10 (b) C3H8 (c) C2H6 (d) CH4
(a) increasing volume (b) applying pressure
(c) increasing temperature (d) replacing pressure
(a) gas carbon (b) coal gas
(c) Biogas (d) marsh gas
(a) the ignition temperature is brought down by sand
(b) supply of air is cut off
(c) sand is a bad conductor of heat
(d) all of these
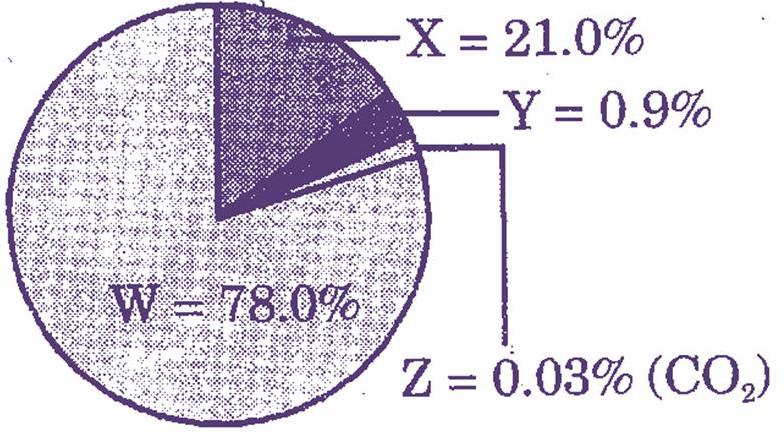
The pie chart above represents the composition of air in its definite proportions. Identify the gases W, X and Y.
(a) W-Nitrogen
X-Rare gases
Y-Oxygen
(b) W-Carbon
X-Oxygen
Y-Rare gases
(c) W-Nitrogen
X-Oxygen
Y-Rare gases
(d) W-Rare gases
X-Hydrogen
Y-Oxygen
Answer Key:
(1)-(a); (2)-(c); (3)-(a); (4)-(a); (5)-(a); (6)-(c); (7)-(c); (8)-(a); (9)-(d); (10)-(b); (11)-(c); (12)-(b); (13)-(c)