Is matter around us pure Worksheet-6
A. Ethyl alcohol and water are miscible liquids whereas oil and water are immiscible liquids
B. Ethyl alcohol and water are immiscible liquids whereas oil and water are miscible liquids
C. The constituents of a homogeneous mixture cannot be separated
D. The constituents of a heterogeneous mixture can be separated
A. Yes, Mixture of ammonia, Hydrogen chloride and ammonium chloride will be formed
B. Mixture and compound both will be formed
C. Homogeneous mixture will be formed
D. No, they will combine to form a new substance Ammonium chloride (compound)
A. Increasing temperature
B. Decreasing temperature
C. Cnetrifugation
D. Increasing the amount of solute
A. Dissolution of Sodium chloride in water is exothermic where as that of Lithium carbonate is endothermic.
B. Heat is released when sodium chloride dissolves in water where as heat is absorbed when Lithium carbonate dissolves in water.
C. Sodium chloride combines with water forming a new compound so its solubility increases with increase in temperature where as solubility of Lithium carbonate decreases with increase in temperature
D. Dissolution of Sodium chloride in water is endothermic where as that of Lithium carbonate is exothermic.
A. Particles of size less than 1nm move fast and collide with each other scattering light
B. Particles of size 1nm to 100nm scatter light
C. Particles of size more than 100nm scatter the light
D. Colloidal solution contains particles of different sizes which scatter light
A. Water forms scum with dirt hence the dirty cloth cannot be cleansed
B. All of these
C. Water dissolves the dirt particles and remains stick to the cloth
D. Soap helps in forming an emulsion between water and oil droplets carrying dust.
A. Fog is (liquid dispersed in gas) and cloud is (solid dispersed in gas)
B. Both are colloidal solutions in which liquid is dispersed in gas but cloud is formed in the upper atmosphere where as fog in the region close to earth
C. Both are colloidal solutions in which liquid is dispersed in gas but cloud is formed in the mesosphere and fog in troposphere
D. Fog is (gas dispersed in liquid) and cloud is (gas dispersed in solid)
A. Distillation
B. Fractional distillation
C. Steam distillation
D. All of these
A. Yes
B. No
C. Depends upon the quantity
D. Can’t say
A. Impurities are present in solid due to which there is difference in melting points
B. There is possibility of error in the measurement of M.P.
C. During the measurement process solid might have reacted with air forming its oxide due to which there is difference in melting points
D. During the measurement process solid might have reacted with air forming its carbonate due to which there is difference in melting points
Answer:
Explanation: Ethyl alcohol (or simply alcohol) and water are miscible. The solution formed is homogeneous i.e. there is no boundary of separation between ethyl alcohol and water. Oil and water are immiscible liquids. The solution formed is heterogeneous. There is a boundary of separation between the two which means that oil and water form separate layers.
Explanation: No, Mixture will not be formed because they combined with each other to form ammonium chloride which is a new substance.
A mixture is always formed by mixing non-reacting substances.
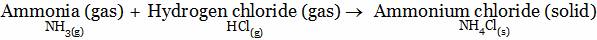
Explanation: A saturated solution can be made unsaturated in two ways:
By increasing the temperature or by heating.
By adding more of the solvent or by diluting the solution.
Explanation: The dissolution of sodium chloride in water is endothermic i.e. heat is absorbed when sodium chloride dissolves in water. Therefore, solubility increases with rise in the temperature.
Dissolution of Lithium carbonate is exothermic i.e. heat is evolved when lithium carbonate dissolves in water. Therefore, solubility of Lithium carbonate in water decreases with rise in temperature.
Explanation: In a colloidal solution, the particle size is such (1 nm to 100 nm), these particles scatter the light rays as they fall on them. Because of scattering, the path of the light as well as the particles becomes visible. But in a true solution, the particle-size is so small (less than 1 nm) that these particles are not in a position to scatter the light. Therefore, true solution does not show any Tyndall effect.
Explanation: In dirty clothes, the dust particles are present on oil drops sticking to them. Simple water cannot remove these oil drops from the clothes because water and oil as such do not form a stable emulsion. Soap plays the role of emulsifier and helps in forming a stable emulsion between the two. This means that soap helps in removing these oil drops along with the dirt sticking to them. The dirty clothes get washed by soap solution.
Explanation: Both fog and cloud are the examples in which liquid is the dispersed phase and gas (air) is the dispersion medium. The only difference between them is that clouds are formed in the upper atmosphere while fog gets formed in the region close to earth.
Explanation: In case the difference in boiling point is less than 20°, the vapours of high boiling liquid will also get condensed along with the low boiling liquid. The liquids can not be separated by simple distillation. Separation of the liquids can be done by fractional distillation.
Explanation: No, it will not happen. In water the two elements chemically combine with each other. Water has properties different from the constituent gases. But in the mixture, Hydrogen and oxygen do not combine chemically and retain their property hence cannot be used to extinguish fire.
Explanation: Since the experimentally determined melting point of the solid is more than the standard value of the melting point, this means that the solid is not in pure state. It has some impurities present. Please note that the purity of a solid can be determined by finding its melting point and comparing it with the standard value.